Please note
This trial is no longer recruiting patients. We hope to add results when they are available.
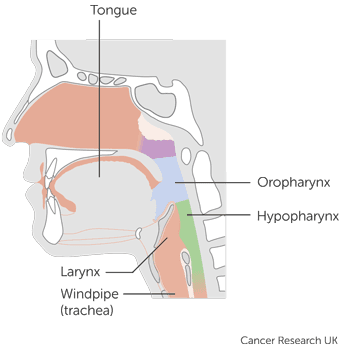
Head and neck cancers, Laryngeal cancer, Mouth and oropharyngeal cancer, Pharyngeal cancer
Closed
Phase 3
This trial is for people with head and neck cancer that has either come back (recurrent cancer) or spread elsewhere (metastatic cancer). It is looking at MEDI4736 alone or MEDI4736 and tremelimumab compared to standard treatment
It is for people with head and neck of the
Recruitment start: 29 February 2016
Recruitment end: 2 March 2017
Please note: In order to join a trial you will need to discuss it with your doctor, unless otherwise specified.
Professor Hisham Mehanna
AstraZeneca
Last reviewed: 08 May 2017
CRUK internal database number: 13751