A trial looking at different ways of giving radiotherapy for cancer of the prostate (PIVOTALboost)
Please note - this trial is no longer recruiting patients. We hope to add results when they are available.
Cancer type:
Status:
Phase:
This trial is to find out if having radiotherapy to the lymph nodes in the pelvis as well as the prostate, improves treatment for prostate cancer. And to see if extra boosts of radiotherapy to the prostate is a useful treatment.
It is for men with a medium to high risk of their cancer coming back.
Cancer Research UK supports this trial.
More about this trial
- which treatment works best and causes the fewest side effects
- if having radiotherapy to the pelvic nodes stops or delays the cancer growing again
- more about quality of life
Who can enter
- have prostate cancer that hasn’t spread elsewhere in the body
- haven’t had treatment yet
- have a PSA that is less than 50 before starting hormone therapy
- are well enough to be up and about for at least half the day (performance status 0, 1 or 2)
- are willing use 2 forms of reliable contraception during the trial and for up to a year afterwards if there is any chance your partner could become pregnant
- are expected to live for more than 5 years
- are at least 18 years old
- have prostate cancer that has gown outside of the prostate gland into nearby tissues and organs in the pelvis (the area between the hip bones), stage T3a, T3b or stage T4
- have a Gleason score of 4 or 5
- have a PSA that is more than 20
- have cancer in more than half of 1 side of the prostate gland or in both sides but it is still inside the prostate gland
- have a Gleason score of 7 (4+3) or above
- have a PSA level between 10 and 20
- have a tumour that is bigger than 6mm or other unfavourable factors that show up in your scans or tissue samples (biopsies) – the trial team will check this
- have cancer that has spread to the lymph nodes, it looks like it might have on a scan or it has spread to other parts of the body
- have already had radiotherapy to the prostate or pelvis
- have had an operation to remove the prostate gland, surrounding tissues, lymph nodes and the tubes that carry semen (seminal vesicles) - a radical prostatectomy.
- have had hormone therapy for more than 6 months when you join the trial. Please note, you might have had hormone therapy for longer than 6 months to delay the start of radiotherapy due to the coronavirus pandemic. If this applies you might be able to join the trial if haven’t had hormone therapy for longer than 12 months.
- have had a chemotherapy drug called docetaxel
- have had both hips replaced or any other implants or metal inside that would make it hard to interpret CT scans or plan radiotherapy
- can’t have an MRI scan for any reason if you are joining certain groups in this trial
- can’t have external radiotherapy to the pelvis for any reason
- are going to have high dose rate brachytherapy (HDR) and you take blood thinning medication that you can’t stop for a short time, you have had your prostate removed and the wound hasn’t healed, you have had a recent blood clot in your leg or lung, you have heart problems or you aren’t suitable to have a general anaesthetic
- have had any other cancer in the last 2 years apart from basal cell skin cancer or squamous cell skin cancer or any other cancer in the past that might need treatment
- aren’t suitable to have radiotherapy because you have inflammatory bowel disease, any other long term bowel problem or problems passing urine
Trial design
- hormone treatment – everyone has this
- MRI scan – to work out which group you go into
- planning of your radiotherapy
- radiotherapy
- high dose brachytherapy (a type of internal radiotherapy)
- boosts of radiotherapy to the tumour area
- boosts of radiotherapy to the whole prostate gland
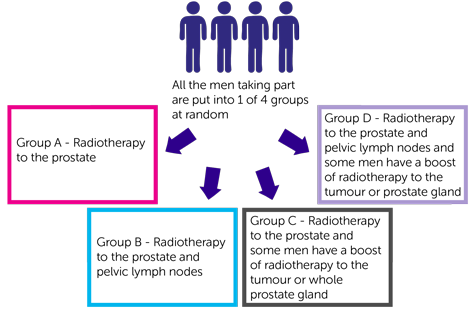
- group A have radiotherapy to the prostate
- group B have radiotherapy to the prostate and pelvic lymph nodes (this group is closed to recruitment)
- group C have radiotherapy to the prostate and some men have a boost of radiotherapy to the tumour or whole prostate gland
- group D have radiotherapy to the prostate and pelvic lymph nodes and some men have a boost of radiotherapy to the tumour or prostate gland
Having intensity modulated radiotherapy (IMRT)
- every weekday, Monday to Friday for 4 weeks
- every weekday, Monday to Friday for 3 weeks
Hospital visits
- blood tests (including a PSA test)
- physical examination
- MRI scan
- every 6 weeks for the first 6 months
- every 3 months up to 2 years
- every 6 months up to 5 years
- once a year after that
Side effects
- tiredness (fatigue)
- frequent bowel movements, loose poo or diarrhoea
- feeling you need to pass urine more often or pain when passing urine
- difficulty getting an erection
- bleeding from your back passage (rectum)
- blood in your urine
- tiredness
- muscle weakness
- hot flushes
- swelling of the breast tissue
- loss of sex drive (loss of libido)
Recruitment start:
Recruitment end:
How to join a clinical trial
Please note: In order to join a trial you will need to discuss it with your doctor, unless otherwise specified.
Chief Investigator
Dr Isabel Syndikus
Supported by
Cancer Research UK
Institute of Cancer Research (ICR)
Other information
This is Cancer Research UK trial number CRUK/16/018.
If you have questions about the trial please contact our cancer information nurses
Freephone 0808 800 4040