A trial looking at chemotherapy and surgery for early bladder cancer (CALIBER)
Cancer type:
Status:
Phase:
This trial looked at different ways of treating early stage bladder cancer that had come back after initial treatment. It looked at surgery and a type of chemotherapy called mitomycin C.
It was for people with the most common type of bladder cancer called transitional cell cancer.
This trial was open for people to join between 2015 and 2017. These results were published in 2020.
More about this trial
Doctors usually treat bladder cancer that hasn’t grown into the muscle wall (early bladder cancer) with surgery. You then might have a type of chemotherapy called mitomycin C. You usually have a single treatment of chemotherapy into the bladder. This is intravesical treatment. Sometimes bladder cancer can come back. And you might need more surgery and further chemotherapy into the bladder.
Researchers thought that a  with mitomycin C but without surgery could be a useful treatment for bladder tumours that had a low risk of coming back. But they weren’t sure how well it worked.
with mitomycin C but without surgery could be a useful treatment for bladder tumours that had a low risk of coming back. But they weren’t sure how well it worked.
In this trial, they wanted to see if mitomycin C without surgery for people whose cancer returned was a useful alternative to surgery.
The aims of this trial were to find out:
- if a course of treatment with mitomycin C alone was a useful treatment for bladder cancer that had returned
- more about the side effects
- how treatment affected
quality of life 
- if enough people would be willing to take part in a
randomised trial 
Summary of results
The trial team found that it was possible to use mitomycin C as a treatment for people with early bladder cancer.
About this trial
This was a phase 2 trial. 82 people took part.
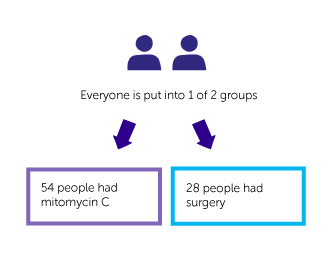
It was a randomised trial. Everyone was put into 1 of 2 groups. Neither they nor their doctor chose which group they were in.
- 54 people had a course of treatment with mitomycin C
- 28 people had surgery
Results
After 3 months treatment the team looked at how many people had no sign of their cancer.
Of the 54 people who had mitomycin C only it was 20 people.
Of the 28 people who had surgery it was 21 people.
The team looked at how many of these people still had no sign of their cancer a year after treatment.
Of the 20 people who had mitomycin C only 15 people did.
Of the 21 people who had surgery 15 people did.
Quality of life
78 out of 82 people took part in the quality of life sub study:
- 51 from the mitomycin C only group
- 27 from the surgery group
The team looked at the quality of life of the people in both groups. They found that it was similar throughout the trial.
Side effects
The team had information about the treatment side effects of 81 people who took part in the trial. The side effects were similar in both groups.
No one reported bad to severe side effects. 14 people reported moderate side effects. 29 people reported mild side effects.
Of the people who had surgery 7 had complications before going home. This was mostly blood in the urine.
Conclusion
The team concluded that to treat early bladder cancer with mitomycin C was possible and safe. Having mitomycin C after surgery might slow the time it took for the cancer to come back than having surgery only. But this isn’t clear yet.
They suggest that further research is needed to find out:
- about whether having mitomycin C before surgery might reduce the chances of cancer coming back
- the best dose to have before surgery
Where this information comes from
We have based this summary on information from the research team. The information they sent us has been reviewed by independent specialists ( ) and published in a medical journal. The figures we quote above were provided by the trial team who did the research. We have not analysed the data ourselves.
) and published in a medical journal. The figures we quote above were provided by the trial team who did the research. We have not analysed the data ourselves.
Recruitment start:
Recruitment end:
How to join a clinical trial
Please note: In order to join a trial you will need to discuss it with your doctor, unless otherwise specified.
Chief Investigator
Mr Hugh Mostafid
Supported by
Experimental Cancer Medicine Centre (ECMC)
The Institute of Cancer Research
NIHR Clinical Research Network: Cancer
NIHR Research for Patient Benefit (RfPB) Programme
If you have questions about the trial please contact our cancer information nurses
Freephone 0808 800 4040